Moving with the power of thought
A device the size of a matchstick, implanted next to the brain's motor cortex, could one day help paralysed people move their limbs
By Jane Gardner
A group of paralysed patients will soon undergo a revolutionary procedure that could allow them to walk with the power of thought.
The key to returning mobility is a tiny, matchstick-sized device called a stentrode. It will be implanted into a blood vessel next to the motor cortex, the brain’s control centre – bypassing the need for complex brain surgery.
From there it will pick up brain signals and allow patients to move a robotic exoskeleton attached to their limbs simply by thinking about it.
This notion of wirelessly thought-controlled limbs is within reach, thanks to a collaboration of 39 brilliant minds from 16 departments across the University of Melbourne’s medicine, science, veterinary science and engineering faculties.
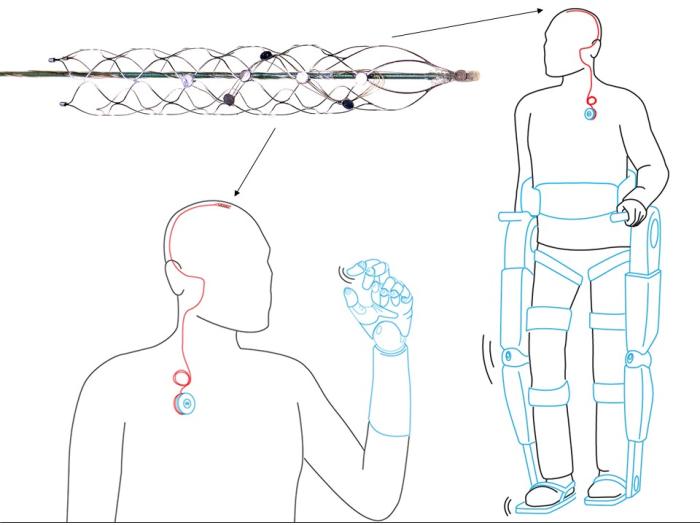

A research paper published in Nature Biotechnology hails the pre-clinical animal trials of the stentrode, which measures only three millimetres wide, a success.
The research team demonstrated the stentrode can pick up strong electrical frequencies emitted by the brain that are coded into a computer. The computer then sends a signal to an exoskeleton attached to the arms or legs, enabling movement.
The work is the result of close collaboration between the University of Melbourne, the Royal Melbourne Hospital and the Florey Institute of Neuroscience and Mental Health.
In late 2017, a select group of paralysed patients from the Royal Melbourne and Austin Hospitals in Australia will be chosen for the trial, where they will be implanted with the stentrode. If the trial succeeds, the technology could become commercially available in as little as six years.

The stentrode could also benefit people with Parkinson’s disease, motor neurone disease, obsessive compulsive disorder and depression and could even predict and manage seizures in epileptic patients.
Neurologist at the University of Melbourne, Florey Institute and the Royal Melbourne Hospital, and current endovascular neurosurgery fellow at Mount Sinai Hospital, New York City, Dr Thomas Oxley, is the founder of this technology.
In 2011, Dr Oxley was travelling in New York, after completing his internal medicine fellowship at The Alfred Hospital, Melbourne.
He had been following the work of DARPA, the US Department of Defense’s research unit, on brain-machine interfaces and decided to cold-call them to pitch a simple yet mind-boggling idea.
What if paraplegics and quadriplegics could power an exoskeleton with their thoughts – without the need for complex and invasive brain surgery?
“I received an email back from Colonel Geoffrey Ling, the military neurologist directing DARPA’s prosthetic limb program, who invited me for an informal chat,” Dr Oxley says. “I put together a sketchy presentation and got on the bus to Washington DC.
“Colonel Ling sat back in his chair and said: go home and put a team together we’ll give you
$1 million to create and test this device.”
Under the supervision of Professor Terry O’Brien, from the Royal Melbourne Hospital and the University of Melbourne’s Medicine, Dentistry and Health Sciences Faculty, Dr Oxley assembled a team of scientists, neuroscientists, surgeons, doctors and engineers.
The DARPA funding was leveraged with two large grants from the Australian National Health and Medical Research Council for a further $2.2 million.
“Professor Terry O’Brien made it happen. He walked me in to the Department of Engineering and introduced me to Tony Burkitt and David Grayden, engineering professors who had worked on the bionic eye and bionic ear projects,” Dr Oxley says.
Professor O’Brien, who is Head of the University’s Department of Medicine at the Royal Melbourne Hospital, has dubbed Dr Oxley’s new technology the ‘Holy Grail’ of medicine.
“This technology is really exciting. It’s the first time that we’ve been able to demonstrate and develop a device that can be implanted without the need for a big operation, to chronically record brain activity,” Professor O’Brien says. “The most obvious benefit is for people who are paralysed following a stroke or spinal cord injury. It is simple and non-invasive and much safer for patients.”
The stentrode, designed in Melbourne and crafted from a space-age alloy called nitinol, will be inserted into the blood vessel with a catheter fed up through the groin – the same approach that has been used for years for cardiology and removing stroke clots.
When the catheter is inserted into the blood vessel in the brain, it leaves a small cigar-shaped ‘basket’, wired with electrodes, which can record the brainwave activity.
“There is no craniotomy, no risk of infection, it’s all run through the groin and passed inside the body up into the brain,” Professor O’Brien says.
“This has been the Holy Grail for research in bionics – a device that can record brainwave activity over long periods. Inside the blood vessel, it’s protected, it doesn’t damage the brain vessel and can stay there forever.”
For Dr Oxley, the situation seemed surreal. He’d gone from having an idea to a multi-million dollar budget and a team within the space of several weeks.
“All of a sudden I’d started my PhD and had several million dollars to work with and a whole area to start my own lab with the engineers,” he says.
Three years of design, development and testing led to the unveiling of an ingenious stent device which can be implanted in a simple day procedure.
Professor Clive May, from the Florey Institute of Neuroscience and Mental Health, is behind trials of the technology in the brains of healthy living sheep.
He recorded this seamless integration of technology and biology, with the device reading electronic brain signals from the motor cortex loud and clear. The sheep were unaffected by the painless, quick and simple operation and were walking and eating within an hour.
“As the device absorbed into the vein wall after nine or so days, the electrical signals continued to become clearer and stronger, up to 190 hertz, as strong as signals previously recorded with intricate invasive brain surgery,” Professor May says.
These signals act as the electrical messengers that provoke intricate muscle movements and can, theoretically, be coded into software that links to an external skeleton.
“Personally, the fact that our device can record signals up to 190 hertz is the most exciting finding in our Nature Biotechnology paper,” Dr Oxley says. “The data between 70 to 200 hertz is the most useful for brain machine interfacing.
“Very smart people told us that we would have big problems with blood creating so much noise it would interfere with the signal, but we showed this isn’t true.”
The chief engineer behind the device is the University of Melbourne’s Dr Nick Opie, a Senior Research Fellow and co-head of the Vascular Bionics Laboratory at the Royal Melbourne Hospital.
He and Dr Oxley co-founded SmartStent, the company that will translate this research into reality. Dr Opie was recruited from the bionic eye project.
“I’ve always been fascinated by the integration of man and machine and the ways that people and machines could function together. Fortunately, I was born in the time to do this,” Dr Opie says.
His challenge: to engineer a tiny net-like device that could be fitted with electrode receivers.
It needed the ability to collapse to a tiny few millimetres in diameter and spring back into shape to act as a scaffold to maintain the flow of blood and permanently settle into the vein. Crucially, the device needed to be biocompatible.
“The first iteration was pretty horrible,” he admits. “I don’t want to count how many we’ve made.
It required a lot of microscope work and very steady hands.”
Hundreds of iterations later, Dr Opie and his team produced the winning design using a flexible material called nitinol, also used in bra underwires and for modern glasses frames.
It is fitted with tiny recording discs, called electrodes, which sit on the wall of the blood vessel, right next to the brain tissue.

Each disc records electrical activity fired by some 10,000 neurons, which is delivered via delicate wires that run out of the brain, into the neck and emerge into the chest into a wireless transmission system.
The researchers say this transmission can be coded into signals that control an exoskeleton. The first patient will work hard to ‘code’ each of these unique signals to their exoskeleton.
Much like the process of learning to walk or speak again, the process will take many months, until finally, the movement becomes as effortless as driving a car, touch-typing, or writing your name on a form.
“Imagine someone bought a piano for you and you didn’t know how to play,” Dr Oxley explains.
“You know that your hands are physically capable of playing it, but you don’t understand the sequence in which the keys have to be struck. It will take time to use your hands to learn how to play the piano. With our device, you’ve essentially connected an electronic limb to the patient’s brain, but they have to learn how to use it.”
Human trials in 2017
In 2017, the stent will be implanted into carefully selected paraplegic or quadriplegic patients by surgeons at the Royal Melbourne Hospital. The long road to coding will begin at the Austin Hospital’s Spinal Cord Service.
The first patients will most likely be young people who have suffered a traumatic spinal cord injury around six months to a year earlier, who are suitable for exoskeleton legs. They will be chosen for their level of determination, their resolve and their physiology, Dr Opie says.
Dr Opie will be there to guide the first patients through the journey. He says one of the remarkable aspects of his device is it does not require opening up the skull to implant.
“Other groups around the world have been looking at an invasive method, where you remove the skull and put electrodes on to the surface of the brain or use a pneumatic gun to fire them into the tissue,” he says.
“There are issues with this. First, when the electrode is directly implanted into the brain it requires very invasive brain surgery, with a 26 per cent risk of bleeding and infection.
“Secondly, the body sets off a series of immune responses that cover the electrodes in scar tissue and stops them functioning.”

Melbourne School of Engineering Professor David Grayden was the senior engineer overseeing the project. He was instrumental in securing the DARPA funding.
Professor Grayden says the stentrode has potential to provide electrical stimulation, as well as receiving signals, and could be useful for the bionic eye.
“In the future, these electrodes could stimulate the brain. It could be used as a complementary stimulation for a bionic eye,” Professor Grayden says.
He says if the retina is not functioning, some vision could be restored by stimulating the brain directly. But the challenge has always been the part of the brain responsible for sight is difficult to get to with surgery.
There are, however, blood vessels that run near that area that could be fitted with the device, without the need to operate on the brain itself. “If we can record a signal coming from the brain, then maybe we can feed signals back in,” he says. “We’re only just starting to extend our research into that area.”
Researchers across all fields
Professor Grayden says the expertise across the University of Melbourne was key to the stentrode project’s success.
“This project shows enormous potential for the development of breakthrough technologies when researchers from engineering, science and medicine work together. The bionic ear was developed here, the bionic eye has been developed by us, so we’ve got a great track record in this area.”
Dr Oxley agrees.
“None of this would’ve happened if I wasn’t with the University of Melbourne. No other institution in the country would be able to pull a project like this off. I was able to go back to DARPA and tell them I had the chief engineer from the bionic eye project on board,” Dr Oxley says.
“Having access to the Florey Institute for Neuroscience and the Royal Melbourne Hospital means you have a huge range of experts to get advice from.”
But, he says, it will be a matter of years before people with paralysis will be able to ask for this treatment.
“The process for getting commercial approval for new medical devices is a long process, so realistically, it could be another five to seven years away,” Dr Oxley says. “And during that five years, we’d have to do a broader clinical trial of closer to 30 to 40 people. So we are hopeful this will be on the market by 2022.”
Multi-media content: Paul Burston and Sarah Fisher.
Banner image: Shutterstock.
This work was funded by DARPA, the NHMRC, the US Department of Defense, US Office of Naval Research Global, the Australian Defence Health Foundation, the Brain Foundation.
This article was first published on Pursuit. Read the original article.

